“Why hasn’t anybody found a cure for cancer yet?” I’ve been asked that question by family members and friends more times than I can count.
It’s an excellent question.
With all the money and effort that has gone into cancer research, why does it feel like we’re not gaining any ground? What’s it going to take to beat this thing?
I wish the answer were simple, but the truth is, cancer is an incredibly complex disease.
To start with, there are many different kinds of cancer, each with its own set of contributing factors. For example, the age of the patient, diet, genetic makeup, tissue of origin and the ability of cancer cells to evolve can drastically affect how and when cancer spreads.
When you look at the problem of cancer in the abstract, it’s so dauntingly complex that the idea of science ever being able to cure it seems frustratingly out of reach.
Things become even more complex when you compare adult cancers to pediatric cancers, which is the area of research my lab focuses on.
Here’s why: When a normal cell becomes a cancer cell, it must acquire a set of “hallmark” characteristics. Some of these characteristics enable the cell to multiply without restriction. Some provide the ability to evade cell death. Some form new blood vessels to carry oxygen and nutrients, which allows the cancer to spread to other tissues.
These are all processes that normal cells need for proper development into tissues, organ systems and so on, but cancer cells take over these processes and use them to their advantage, which can be dangerous.
What remains unclear is why these cells suddenly become cancer cells. Historically, it is thought that these “hallmarks” are acquired through multiple mutations within genes encoded in the DNA, a type of molecule that acts as the instructions for a cell. Basically, the instructions are corrupted, resulting in the formation of cancer.
The above is true for many adult cancers, but studies over the last 10 years have revealed that this is not the case for some pediatric cancers.
These cancers have far fewer mutations and, in a small subset, just one mutation is found. This is highly unusual and leads to an interesting question: Why would those pediatric cancers form if they don’t acquire as many mutations as adult cancers? What else could be happening?
Well, others and I have learned that there are additional mechanisms, beyond mutations in genes, that cancer cells can use.
One example of an additional mechanism is epigenetics, which is defined by groups of different proteins that essentially affect how the gene is read.
Think of it as the instructions in a foreign language, and you are trying to translate them as accurately as possible. The instructions are correct, but it’s the translation that is flawed.
In fact, in my earlier studies in retinoblastoma, a childhood cancer of the eye, we identified epigenetic changes in tumor cells that led to the activation of many cancer-causing genes that are supposed to be turned off.
That research led to exciting progress: We were able to identify a new drug to shut down one of these cancer-causing genes, meaning it can be used as a potential therapy.
By exploring these non-genetic mechanisms, we were able to unearth new therapeutics for retinoblastoma.
Fortunately, after decades of research, retinoblastoma is now curable with more than 95 percent of patients surviving.
However, survival rates for other pediatric cancers are much lower and have not improved for more than 30 years. One of these cancers is called rhabdomyosarcoma, which is a cancer of skeletal muscle cells. Like retinoblastoma, a subset of patients has a type of rhabdomyosarcoma that carries only one major mutation known as the PAX3/FOXO1 translocation. This mutation is essentially the parts of two genes stitched together to make a whole new gene.
To date, it is not entirely clear how this translocation is turned on. It is also not clear what other mechanisms contribute to developing rhabdomyosarcoma besides this one mutation.
These questions about rhabdomyosarcoma are what my lab and our collaborators are focused on answering right now.
In fact, we discovered a new non-genetic mechanism that turns on this translocation and also helps to keep the cancer cells alive.
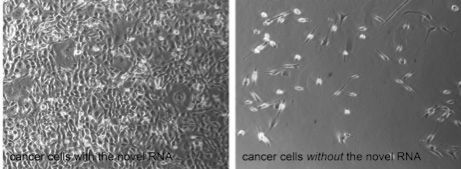
This mechanism is mediated by a class of novel RNAs, molecules that act like a second set of instructions provided by the DNA.
As shown in the accompanying image, we removed one of these RNAs in rhabdomyosarcoma cancer cell lines and observed rapid cell death.
In addition, we found that the PAX3/FOXO1 is no longer turned on and, in contrast, genes that normally function to suppress cancer formation are now turned back on.
This is a remarkable finding because not only have we identified a regulator of the PAX3/FOXO1 gene, but we also discovered a novel mechanism that drives this cancer.
Our lab is currently working on how these RNAs function and what other genes they regulate.
We expect that these data will open new opportunities to explore therapeutics that haven’t been considered for treating children with rhabdomyosarcoma. Research like this could significantly impact the longevity and quality of patients’ lives. And that, hard and time-consuming as it is, is real progress.
So, here’s what I tell my family and friends when they ask me what’s taking so long: Every day, inch by inch, doctors and researchers all over the world are working steadily toward the treatment and cure of many different types of cancer.
And with every new piece of the puzzle we begin to understand, our dream of curing all forms of cancer moves that much closer to reality.
We just have to keep at it.